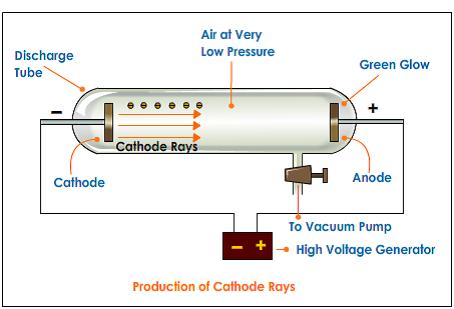
To illustrate the effects of an electric field on a charged particle.To commemorate the discovery of the electron by J.As a result of his work, Thomson proposed a completely new model of the atom (Thomson’s Plum Pudding Model) that one of his students, Ernest Rutherford, would improve upon 10 years later.Ĭlick and drag the cursor to change the intensity of the voltage applied between the plates. This apparatus constitutes the first particle accelerator. Thomson, in 1897, isolated a new elementary particle carrying a negative charge – the electron. The very intense electric field that results from this accelerates the few ions present in the tube which, via collisions, ionize other particles. The lower the pressure, the more the electrons thus liberated and accelerated travel great distances until they strike the screen at the opposite end of the tube.īy studying the deviation of this beam, J. Thomson first investigated the magnetic deflection of cathode rays. A high voltage (between 10 and 100 kV) is applied between two electrodes. J. A refined version of Perrin’s experiment is the centerpiece of Thomson’s talk of April 30, 1897. Thomson’s second experiment involving the deviation of an electron beam in a vacuum tube, called a Crookes Tube.Ī partial vacuum (less than 10 -6 atm) is maintained in the tube. Thomson, a scientist, began working with cathode ray tubes in the late 1800s. reported an experiment in which, contrary to Hertz, the negative electric charge does accompany the cathode rays 11.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
